Dr. Philip McMillan, John McMillan
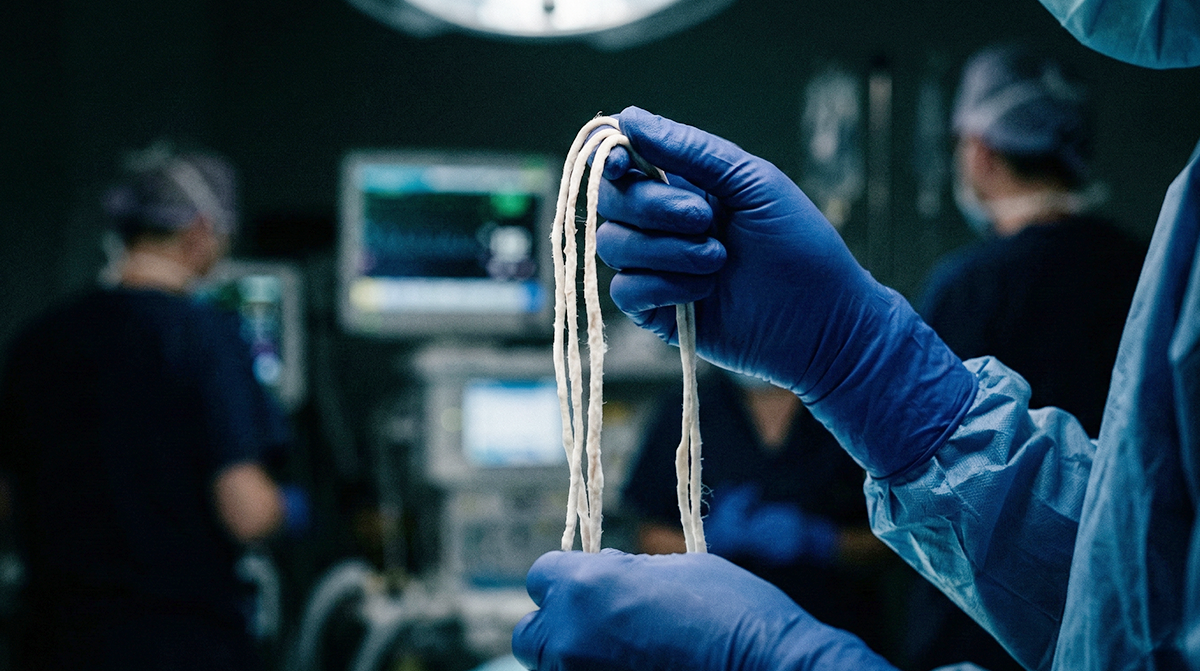
Something strange started showing up on embalming tables around 2021. Long, white, rubbery structures, unlike anything veteran embalmers had pulled from cadavers before, began appearing in blood vessels with unsettling regularity. They weren’t the familiar dark-red, gelatinous clots that mortuary professionals had handled for decades. These were different. Pale, fibrous, and disturbingly large, they looked almost artificial. And they kept turning up.
Dr. Philip McMillan, a researcher and clinician who has spent considerable time investigating this phenomenon, draws a pointed analogy. Imagine, he suggests, that hairdressers, while washing and combing their clients’ hair, start noticing unusual moles on scalps. Some of those moles turn out to be melanoma. Would the scientific community dismiss the observation simply because hairdressers aren’t dermatologists? The embalmers were doing much the same thing: making an observation, raising a question. They weren’t claiming to have the answer. They were asking why nobody else seemed to be looking.
Now, years later, the looking has begun in earnest. Researchers Bruce Rapley and Matt Sheldon have published three papers characterizing the proteomic makeup of these anomalous intravascular casts, the scientific term for what many have colloquially called “white clots.” Their work, conducted quietly over several years, is finally reaching the broader scientific conversation. And the findings are raising questions that the medical community can no longer afford to sidestep.
The Physical Evidence
To appreciate why these clots have generated such concern, it helps to understand what a normal blood clot looks like. A typical clot is red, that deep, dark, grape-jelly red that comes from being packed with red blood cells. It is soft and breaks apart relatively easily under standard anticoagulation therapy. Doctors encounter them routinely, and the protocols for dealing with them are well established.
The white clots are a different beast altogether. Stripped of red blood cells, they are pale and rubbery, with a tensile strength that makes them stubbornly resistant to conventional treatments. Embalmer Richard Hirschman pulled a 19-inch specimen from a single cadaver, white all the way through. A whistleblower sent Dr. McMillan an image of a clot removed from a living patient’s inferior vena cava, the large vein that carries blood back to the heart. The clot was, in Dr. McMillan’s own word, “humongous.” The patient survived only because surgeons performed an emergency embolectomy to extract it.
But the massive clots, dramatic as they are, represent only the extreme end of a spectrum. A smaller version of the same clot could lodge in a kidney, block an artery feeding the leg, travel to the spleen, or trigger a stroke. It could cause a pulmonary embolus that a patient barely survives. The clinical presentation would differ depending on where the clot ends up and how large it is, but the underlying mechanism, if these clots share a common origin, could be the same.
The Proteomic Puzzle: What’s Inside These Clots?
Rapley and Sheldon’s proteomic analysis examined 541 proteins found within the anomalous clots. Two proteins dominated: hemoglobin subunit beta and fibrinogen beta. That hemoglobin showed up at all was telling, it meant red blood cells were being destroyed and their contents incorporated into the clot matrix. But the really puzzling detail was the ratio.
Normal hemoglobin is built from two alpha chains and two beta chains in equal proportion. Inside these white clots, the beta subunit was present in significantly higher quantities than the alpha chain. That imbalance nagged at Dr. McMillan for some time. If hemoglobin was being liberated from broken-down red blood cells, why weren’t both subunits appearing in roughly equal amounts? Something was selectively stripping the hemoglobin apart and leaving the pieces unevenly distributed.
It is a detail that might seem arcane to anyone outside haematology, but it matters enormously. The imbalance is a fingerprint, a clue pointing toward whatever biological mechanism is driving the formation of these clots. And it was this specific anomaly, this lopsided ratio of beta to alpha, that eventually led Dr. McMillan toward a hypothesis he had not anticipated.
The Breakthrough Hypothesis: G6PD Deficiency
While researching the role of a specific immune cell, a THBS monocyte, in the rapid breakdown of red blood cells and the removal of free hemoglobin, Dr. McMillan stumbled across a connection he hadn’t been looking for. Glucose-6-phosphate dehydrogenase deficiency, commonly known as G6PD deficiency, is an inherited condition that affects more than 400 million people worldwide, with the highest prevalence in African and Middle Eastern populations.
In people with G6PD deficiency, red blood cells are unusually fragile. When the body is placed under stress, by infection, by certain medications such as hydroxychloroquine, or even by something as mundane as eating fava beans, those red blood cells can rupture, releasing their hemoglobin contents, including both alpha and beta chains. Critically, individuals with G6PD deficiency also tend to be more susceptible to inflammatory triggers, which could push them toward greater thrombosis and clot formation.
Dr. McMillan is cautious to frame this as a hypothesis rather than a conclusion. But if the connection holds, the implications are significant. It could mean that a definable cohort of patients, those with G6PD deficiency, are at disproportionately higher risk for these unusual thrombotic events. And if that cohort can be identified, it opens the door to screening, early intervention, and targeted treatment strategies.
The Clinical Crisis: Why This Cannot Be Ignored
One of the more alarming aspects of the white clot phenomenon is how quickly these structures seem to form. Angiograms, imaging procedures in which dye is injected into blood vessels, are performed every day in hospitals around the world, and these clots are not routinely visible. That suggests they are not growing slowly over weeks or months. Dr. McMillan’s clinical suspicion is that they accumulate rapidly, possibly within days, driven by some unusual and as-yet poorly understood mechanism.
Equally troubling is the resistance these clots show to standard anticoagulation. Normal blood clots can typically be managed, even dissolved, with well-established drug regimens. White clots, by contrast, do not appear to break down under the same treatment. For clinicians, this poses a stark problem: if a patient presents with a clot that does not respond to the usual protocols, what then? The answer, for now, is uncomfortably unclear.
“If it is happening, and you are ignoring it, what are the likely disease presentations that will occur if clots are forming that are very difficult for the body to break down, and additionally, won’t easily be broken down, even with anticoagulation?”
That question, posed by Dr. McMillan, cuts to the heart of the matter. The patients with massive clots, the ones who end up on operating tables or in the morgue, are impossible to miss. But what about the patients whose clots are smaller, whose symptoms mimic a dozen other conditions? A stroke attributed to conventional atherosclerosis. A pulmonary embolism chalked up to immobility. A blocked renal artery assumed to be a one-off event. If white clots are behind even a fraction of these presentations, the current diagnostic framework may be missing them entirely.
Science Demands Answers, Not Beliefs
Dr. McMillan is blunt about what he sees as the greatest obstacle to progress on this front: ideology masquerading as scepticism. The reflex to dismiss uncomfortable findings because they don’t align with prevailing narratives is, he argues, the antithesis of the scientific method.
“Nobody cares what anybody believes. The question is, is it real? And if it’s real, what do you do about it?”
The clots are real, that much is no longer in dispute. They have been pulled from cadavers and from living patients. They have been subjected to proteomic analysis across three published papers. They contain unusual patterns of proteins that do not match the composition of ordinary blood clots. And they resist conventional treatment.
What remains is the harder work: understanding why they form, who is most vulnerable, and what can be done. The G6PD hypothesis is one promising thread, but it is just that, a thread. It needs testing, replication, and scrutiny. The beta-to-alpha hemoglobin imbalance needs a mechanistic explanation that holds up under controlled conditions.
Science, at its best, is observation followed by inquiry. Something appeared on embalming tables that had not been there before. Researchers picked it up, examined it, and published their findings. The medical community now faces a choice: pursue the questions these findings raise, or look away and hope the problem resolves itself. For the patients whose lives may depend on the answer, looking away is not an option.


Could this be caused by parasites. Has there been any findings or research.